
The GRPs proteins are expressed constitutively 
Several protein kinases to up-regulate the protein-folding capacity of the ER. In the mid-1980s, it was shown that BiP is identical to GRP78 ( Munro and Pelham 1986), the 78-kD family member of GRPs that has homology to the cytosolic Hsp70 stress protein.Īs a protein-folding compartment the ER is exquisitely sensitive to alterations in homeostasis. Because their induced expression was a consequence of glucose deprivation, this protein family was named the glucose-regulated Independently, BiP was identifiedĪs a member of a protein family that is expressed at a high level in virally transformed cells and upon conditions of glucoseĭeprivation ( Lee 1987). In this manner, BiP prevents the secretion of incompletely assembled immunoglobulins. BiP was identifiedĪs an ER-localized protein that bound to heavy-chain immunoglobulins and inhibited their secretion in the absence of lightĬhains in pre-B lymphocytes ( Haas and Wabl 1983). One of the best-characterized examples of this latter class is the immunoglobulin binding protein BiP. These proteins prevent protein-folding intermediates from aggregatingĪnd stabilize energetically unfavorable conformations of polypeptides to minimize irreversible dead-end protein misfolding Second, there are protein chaperones that do not actively catalyze protein folding,īut rather maintain proteins in a folding-competent state. First, there are proteins such as protein disulfide isomerase (PDI) and cis–trans prolyl isomerase that catalyze protein-folding reactions and increase the rate, without changing the pathway, at which proteinsĪttain their final folded conformation. These chaperones provide two important functions in protein folding ( Gething and Sambrook 1992). The ER provides a unique oxidizing environment in which reside numerous protein chaperones facilitate and promote the productiveįolding of proteins and protein complexes despite the presence of high concentrations of protein that would otherwise interfere Next Section The ER is a protein-folding compartment Mechanisms that coordinate the complex ER stress response at the translational and transcriptional level in yeast and in higher The purpose of this review is to summarize what is presently known about the diversity of molecular signaling Recent evidence supports findings that these signaling pathways influence pathogenesis associated with viral infectionĪnd genetic disease. 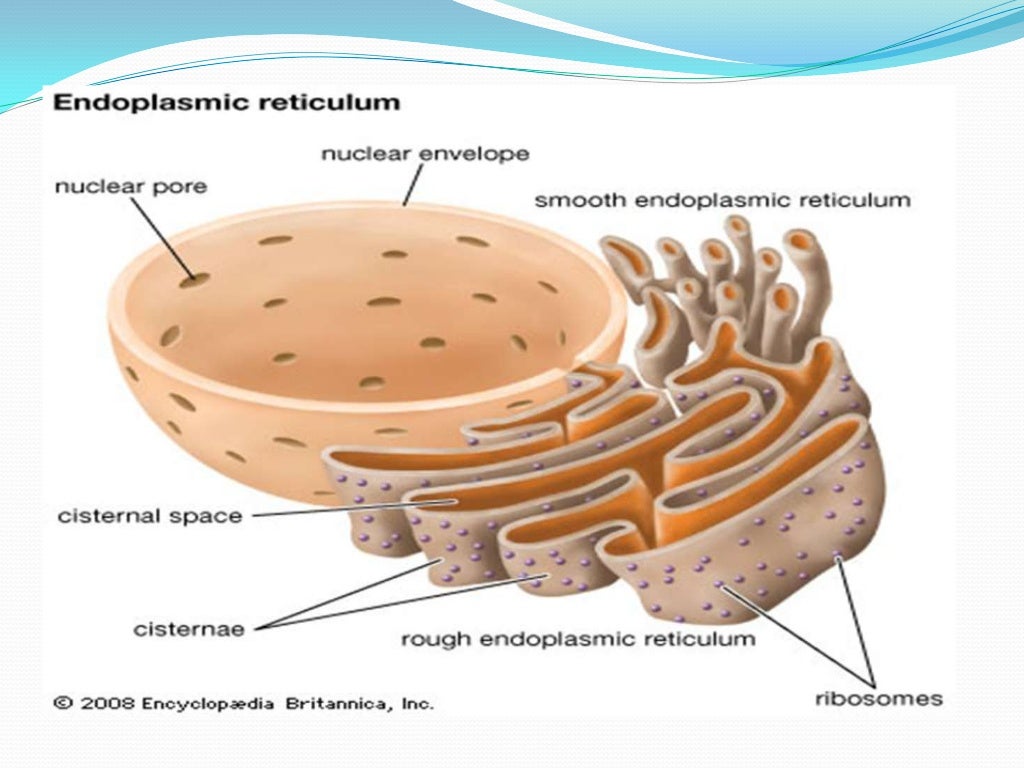
Response occurs at the translational apparatus, whereas changes in gene expression promote long-term adaptation or apoptoticĬell death. The ER to the cytoplasm and the nucleus to eventually result in adaptation for survival or induction of apoptosis. The ER isĮxquisitely sensitive to alterations in homeostasis, where, upon a variety of different stimuli, signals are transduced from Signal-transducing organelle within the cell that continuously responds to environmental cues to release calcium. The ER is also the site of synthesis of cellular lipids and sterols. Proteins are chaperones and catalysts of protein folding that form a matrix on which newly synthesized proteins attain theirįinal conformation. Their continuous vesicle-mediated retrieval from post-ER compartments within the early secretory pathway. Numerous cellular proteins reside within the ER through a mechanism that requires Such as lysosomes and the Golgi compartment. The ER provides a unique oxidizing compartment for the folding of membraneĪnd secretory proteins that are destined to the cell surface, as well as for proteins destined to other intracellular organelles, Modification, folding, and oligomerization occurs. Approximately one-third of all cellular proteins are translocated into the lumen of the ER where post-translational Department of Biological Chemistry and Howard Hughes Medical Institute, The University of Michigan Medical Center, Ann Arbor,Īll eukaryotic cells have an extensive membranous labyrinth network of branching tubules and flattened sacs called the endoplasmic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
